
csv file must be accurate for the icon to be loaded and displayed. The partial pressure of ammonia (A) in air-ammonia mixtures in equilibrium with their aqueous solutions. It should be noted that the image path/filename given in the. can operate (still have vapor and liquid present)DePriester chart for equilibrium data (Wankat, 2007) 3. If new images are added, they should be placed within the /img directory. The image value can left blank without consequence, and will just result in no image being displayed for that compound. (Please note the coefficeint examples ARE NOT actual values) There are a total of 13 entries per compound in the. Each compound is located on a new line (row) in the. command: java -jar kcalc.jar path/m圜ompoundData.csv) Adding New CompoundsĪdding new compounds can be trivially done by editing the. csv file that is not the default, simply call the program from terminal using the. To run the program using compound information from a. jar file is located in, there must be a directory called img, in which all compound icons are located in. csv path and file name.Ībove the directory the. jar without arguments through the terminal, it will launch using the default. csv file, from which all the compound names, coefficients, and icons paths are. DePriester in an article in Chemical Engineering Progress in 1953. jar can be called from terminal using the standard command: java -jar kcalc.jar. DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature. Understanding the concept behind Flash Distillation, the vapor-liquid equilibrium. It is also one of the most important processes to learn in Mass Transfer / Separation Process Technologies as it is a fundamental unit operation. DePriester Chart for a given P, find Tbp (i.e., K 1) for a given T, find Psat (i.e., K 1) for a given P, T, find K K > 1 prefers vapor phase K < 1 prefers liquid phase Don’t extrapolate beyond the range of the chart.
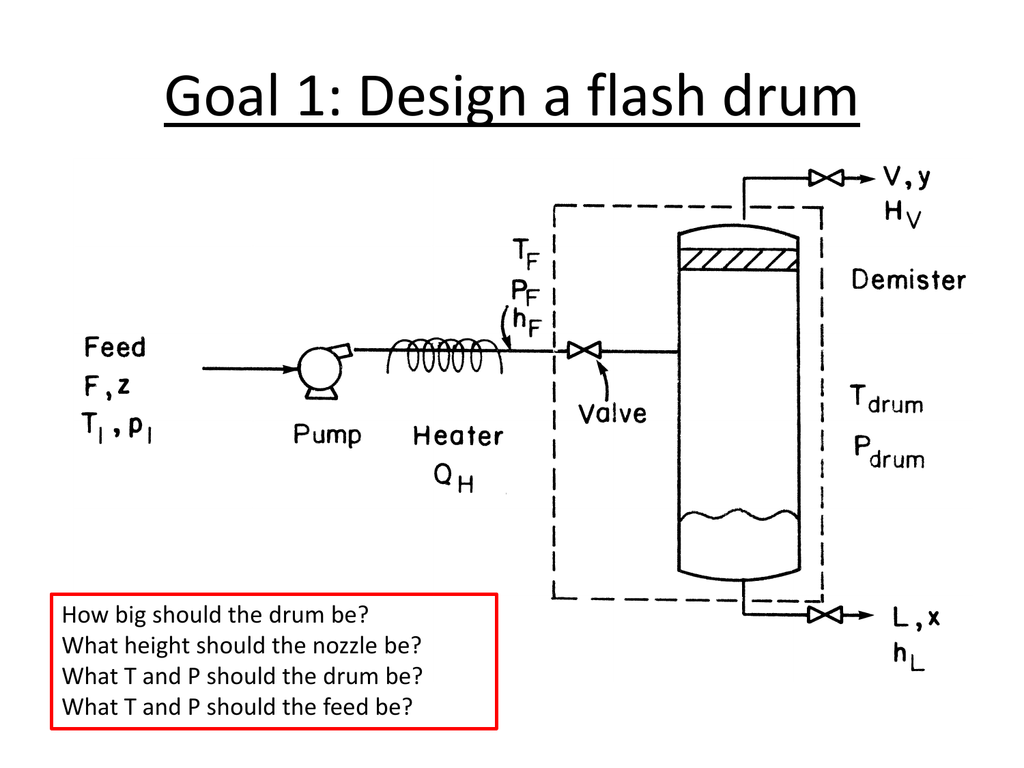
jar can be run by double clicking on the icon from a file explorer. Flash Distillation is one of the most important Mass Transfer Operations used extensively in the Chemical industry. from Introduction to Chemical Engineering Thermodynamics (7th ed) by Smith, J.M., Van. This calculator essentially acts as a DePrieseter chart, calculating the vapor-liquid equilibrium ratio (ie K-Value) for a variety of compounds at a particular temperature and pressure.
